Urine sampling: A way to continue cervical cancer screening in the face of a pandemic
You are here

Cancer screening rates have dramatically dropped due to lockdown
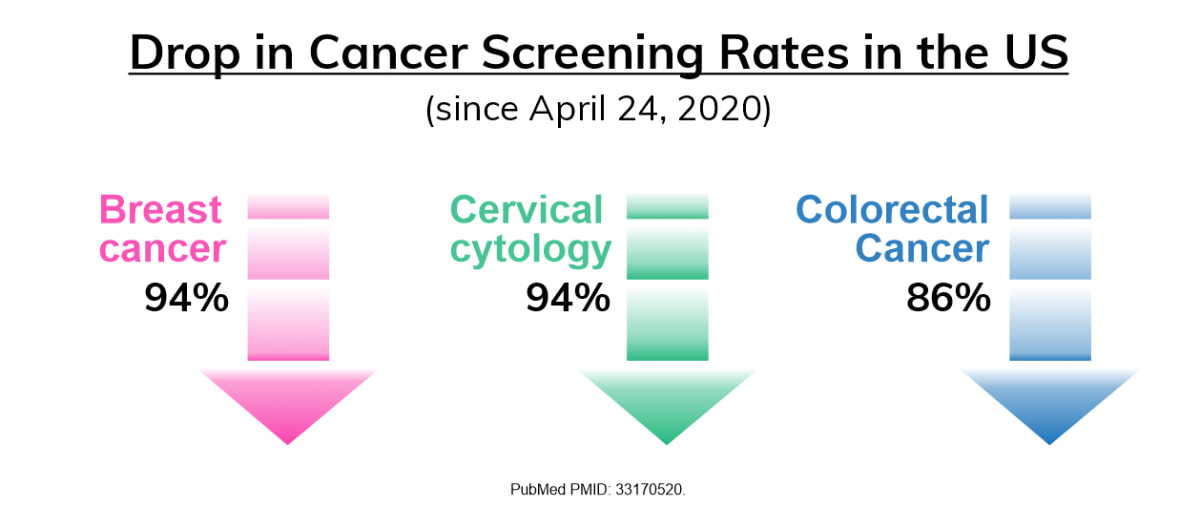 The world has changed in many ways this year; healthcare systems have been under pressure; hospitals have reached capacity in many areas and patients have postponed visits and routine checkups to healthcare facilities and physician offices due to the fear of COVID-19. One area that has been significantly hit due to the pandemic is cancer screening. In the United States (US), for example, as of April 25, 2020, a 94% drop was seen in cervical cytology and breast cancer screening, followed by an 86% drop in colorectal cancer screening (1).
The world has changed in many ways this year; healthcare systems have been under pressure; hospitals have reached capacity in many areas and patients have postponed visits and routine checkups to healthcare facilities and physician offices due to the fear of COVID-19. One area that has been significantly hit due to the pandemic is cancer screening. In the United States (US), for example, as of April 25, 2020, a 94% drop was seen in cervical cytology and breast cancer screening, followed by an 86% drop in colorectal cancer screening (1).
In the United Kingdom (UK), COVID-19 has put huge pressure on cancer services, including screening, testing and treatment. Backlogs have caused delays to screen, with around 2.1 million people waiting for breast, bowel or cervical screening. In many areas of the UK, screening programs have also been suspended due to lockdown, resulting in long-term implications. For every week that these programs are paused, 380 potential cancers are not being diagnosed (2).

Alternative screening methods are necessary to achieve elimination of cervical cancer
Consequently, finding alternative methods to continue cancer screening is necessary to save lives. Additionally, to achieve the World Health Organizations’ (WHO) recently launched global strategy to accelerate the elimination of cervical cancer by 2030 , continuing screening is critical, even during the pandemic. Regular cytology-based screening for cervical cancer through a Pap smear requires a clinician to perform, which can be difficult in the current situation. The new imperatives support self-collection techniques for Human Papillomavirus (HPV) testing, as the primary cervical screening method (3). HPV is responsible for most cervical cancer cases. HPV-based screening provides 60-70% greater protection against invasive cervical carcinomas compared with cytology (4). It also allows for self-sampling, which has proven to be effective in increasing participation and screening coverage (5).
Urine as a sample type can improve cervical cancer screening rates and allow for home-collection
Women have shown a significantly higher preference for urine as a sample type for HPV testing in comparison to brush-based cervical-vaginal self-sampling methods (6). Similar sensitivities have been observed between urine and clinician-taken smears as well as brush-based self-sampling for detecting high-grade precancer lesions i.e. CIN2+ (cervical intraepithelial neoplasia grade 2+), highlighting the potential of urine (7). Urine as a sample is also beneficial as it allows easy, non-invasive collection, which can be performed at home, offering a solution to avoid the disruption of preventive services. However, without the use of an appropriate preservative, as well as control of time and temperature, the quality of the urine sample may be affected during transport from home to the laboratory, affecting diagnostic accuracy.

Read our blog about at-home urine sampling
Download our white paper – Storage and transport recommendations
Colli-Pee® allows easy first-void urine collection and outperforms a regular urine cup
Interestingly, not all urine fractions are the same. Studies have shown that first-void urine (initial urine flow) contains higher concentrations of HPV DNA than midstream urine fractions (8). The first-void fraction contains washed away mucus and debris from exfoliated superficial cell layers of a cervix carcinoma. Therefore, collecting this part of urine is important for improved sensitivity.Collecting a first-void urine sample with a standard urine cup can be awkward, messy and inconvenient for the user. For this reason, Novosanis developed Colli-Pee®, a first-void urine self-sampling device. Colli-Pee® usability data show more accurate and volumetric collection of first-void urine than a traditional urine cup (9).
Additionally, the device architecture also enables immediate mixing of the sample with a urine preservative, improving the stability of the sample (8). Colli-Pee®, prefilled with non-toxic Novosanis proprietary UCM is recommended for HPV testing to ensure stabilization of urinary content .
As Colli-Pee® prefilled with UCM allows for transport at room temperature, it is suited for urine collection at home and postal delivery, which could be a way to avoid unnecessary travel to the hospital and continue screening programs during a pandemic.
Conclusion:
Despite the benefits, cervical cancer screening methods have drawbacks, resulting in poor attendance. This has increased even further because of the pandemic. Alternative sampling methods that are easy to perform independently are essential to continue healthcare practices. Urine as a sample type has shown merit in this area, and given its ability to detect HPV, has the potential to reach more women for cervical cancer screening, especially in these challenging times.
References:
(1) PMID: 33170520
(2) https://scienceblog.cancerresearchuk.org/2020/06/01/impact-of-coronaviru...
(3) PMID: 33072179
(4) PMID: 24192252
(5) PMID: 23618210
(6) PMID: 28689156
(7) PMID: 28391609
(8) PMID: 30452931
(9) https://novosanis.com/sites/default/files/poster/pdf/Meers%20et%20al_Eur...
