VOCs in urine: Novel biomarkers in cancer research?
You are here

Background:
Worldwide cancer is a leading cause of death. By 2040, 29.5 million new cancer cases are estimated, compared to 18.1 million cases reported worldwide in 2018 (1). Early diagnosis and treatment have the potential to improve the quality of care and reduce mortality. However, to screen and reach more individuals, reliable patient-friendly diagnostic methods are required.
Volatile organic compounds (VOCs):
Cancer biomarkers found in tissues as well as bodily fluids, can be used for early detection as well as monitor disease progression. A high number of potentially informative cancer biomarkers, including DNA, RNA, proteins and metabolites have been found (1). Recent research has shown that volatile organic compounds (VOCs), a group of small molecule metabolites, that are continuously generated in the human body and released through bodily fluids such as breath, blood, skin, urine and fecal samples can act as biomarkers in cancer detection. Distinct urinary VOC patterns have been associated with different cancer types, as well as cancer stages (2).
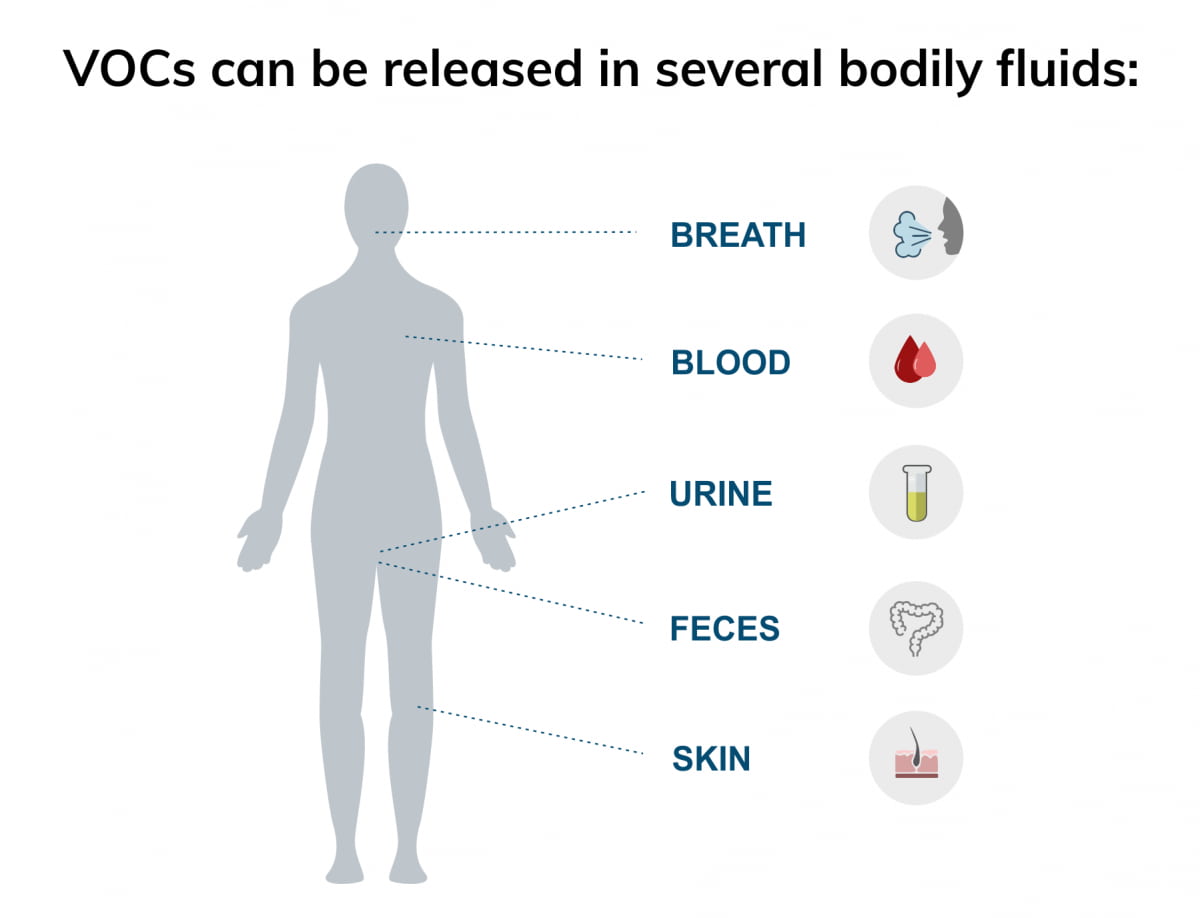
VOCs are easy to detect by analytical instruments, such as gas chromatography-mass spectrometry (GC-MS) as well as highly developed gas sensors. Further, urinary VOCs are interesting as urine can contain cells, proteins, and metabolites originating from the kidney, prostate, and bladder, allowing detection and monitoring of various urological cancers (2). A recent review showed that urinary VOC detection has the potential to be used as a safe, noninvasive, and specific test method for the early detection of urological cancers including bladder, prostate and almost all types of renal cancer (2). VOCs are not only limited to urological cancers but can also be used in breast (3), colorectal (4) and pancreatic cancer research (5). As most pancreatic cancers are diagnosed at an advanced stage, prognosis of this cancer type remains poor. Consequently, earlier detection can help achieve a better prognosis. A proof-of-concept study indicated that urinary VOC detection distinguished between patients with early stage pancreatic cancer from healthy controls (5).

Urine as a sample type:
Urine is easily accessible, non-invasive, available in larger quantities and applicable for home collection. Moreover, the collection does not entail any risk of transmission of blood-borne pathogens (1). Given the benefits of urine sampling, urinary VOC detection for cancer research is attractive.
A study on a small population showed the use of first-void morning urine for the detection of VOC biomarkers in bladder cancer patients (6). Here, researchers aimed to develop an optical sensorybased system, with the goal to become a point-of-care diagnostic test, for urinary VOC biomarker detection of bladder cancer. Four urinary VOCs were identified including, ethylbenzene, hexanal, laurie aldehyde and nonanoyl chloride (6).
Interestingly, urine has been well-researched in other oncology-related fields as well. For example, strains of Human Papillomavirus (HPV) are known to cause majority of cervical cancers as well as precancerous cervical lesions (7). Studies have shown that HPV can be detected in urine. HPV DNA is found in higher concentrations in first-void urine (first 20 ml of urine flow) than subsequent fractions as cervicovaginal discharge is released during urine initiation (8). Further, specific analytes for prostate cancer have also been identified in first-void urine (9).
Novosanis’ Colli-Pee® platform consists of distinct variants capturing a range of first-void urine volumes (4 mL to 45 mL)* in polypropylene collector tubes, offering improved diagnostic accuracy and patient comfort compared to a regular urine cup.
To know more about urine terminology read our blog
To know more about urine as a liquid biopsy for cancer detection read our white papers

An initial literature study revealed that polypropylene (PP) can be used as a container material for urine samples to detect VOC as biomarker. Therefore, the Colli-Pee® platform, offers promise for this novel application (10,11,12).
*Some Novosanis products are in development or not available in all geographic regions. Contact us to know the registration status in your region.
References:
- https://novosanis.com/sites/default/files/poster/pdf/White%20Paper%20PCa-version2020-06A_EN.pdf
- PubMed PMID: 31511830
- PubMed PMID: 30792417
- PubMed PMID: 26752703
- PubMed PMID: 30591442
- PubMed PMID: 30168296
- https://www.who.int/news-room/factsheets/detail/human-papillomavirus-(hpv)-and-cervical-cancer
- PubMed PMID: 30452931
- PubMed PMID: 30129068
- PubMed PMID: 27311952
- PubMed PMID: 20591483
- PubMed PMID: 31477781
