At-home urine sampling: A way to continue healthcare practices to prevent the next global health crisis
You are here

At-home urine sampling: A way to continue healthcare practices to prevent the next global health crisis
COVID-19 disrupted important cancer screening
The COVID-19 pandemic has brought several challenges to healthcare. Lockdowns and fear of getting the infection caused many patients to cancel or delay visits and routine checkups. One area that has been significantly hit by the pandemic is cancer screening and care [1].
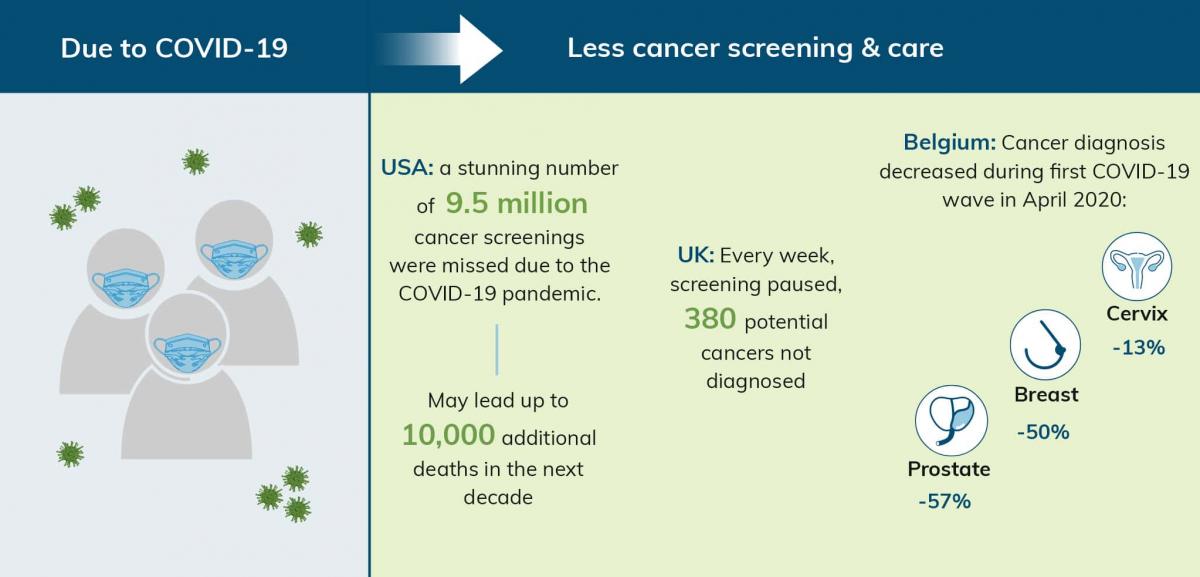
A recent survey from the American Cancer Society Cancer Action Network shows that more than three-quarters of people who were being treated for cancer, delayed some aspect of their medical care due to challenges faced by COVID-19 [2]. The impact was not just limited to those undergoing treatment. The pandemic has also had a huge effect on screening and delayed the diagnosis of cancers such as breast, cervix, prostate, and colorectal cancers [1,3].
For example, the US National Cancer Institute warned drops in screening for breast and colorectal cancer alone may lead up to 10,000 additional deaths in the next decade [2]. Additionally, in the UK, for every week screening programs were paused, 380 potential cancers were not being diagnosed [4]. Further, in Belgium skin cancer diagnosis decreased by 65% during the first COVID-19 wave in April 2020 and is still lagging ever after establishment of several recovery programs [5].
Alternative screening methods are necessary to improve cancer screening and care
Despite the many advances made in cancer screening and diagnostic technologies over the years, the global instability caused by COVID-19 will have long-term implications on cancer mortality rates [2]. To prepare for the future and continue healthcare practices even in the face of pandemic, finding alternative methods to continue healthcare practices such as cancer screening is critical.
Urine as a sample type has shown potential for detecting and monitoring urological and systemic cancers. Additionally, urine is easily accessible, non-invasive, available in larger quantities and applicable for home collection, making the sample type attractive, especially at a time where services are under pressure [5].

However, to use urine for clinical applications, it is important to limit preanalytical errors caused by collection, transport and storage. Studies have shown that not all urine fractions are the same; for example, first-void urine (initial urine flow) contains higher concentrations of several biomarkers than midstream urine fractions.

Novosanis' urine collection device, Colli-Pee® allows for standardized and volumetric collection of urine and ensures immediate mixing with preservative, improving sample collection for downstream analysis [6].
Urine as a liquid biopsy in cancer research
Several biomarker candidates have been identified in urine, including DNA, RNA, proteins, exosomes and metabolites. For different cancer types, detection of biomarkers in alternative sample types such as urine may help continue cancer screening in the face of a pandemic and beyond. Below we discuss a few cancer types where biomarkers in urine have been identified.
-
Cervical cancer
Strains of Human Papillomavirus (HPV), particularly HPV16 and 18 are associated to cause most cervical cancer cases. Cervical cancer usually does not present any symptoms until it reaches an advanced stage, making early detection and treatment challenging.
While cervical cancer screening programs through cytology-based Pap smears have been established in many countries, participation remains low. Many women are often reluctant to undergo screening as the process is invasive and associated with physical discomfort [7].
Self-collection techniques have been recommended to improve screening coverage. HPV can be found in urine, particularly first-void urine (first 20-30 mL of urine flow). HPV-based urine sampling for cervical cancer can help increase participation and reach women that traditionally do not participate in screening programs [7]. Several studies have used Colli-Pee® collected urine for HPV detection for cervical cancer. Some of the publications and results are part of large clinical studies including EVAH, Predictors5.1 and VALHUDES [6].
-
Prostate cancer
Current screening techniques for prostate cancer are focused on serum prostate specific antigen (PSA) levels. Drawbacks of PSA testing are its low specificity, as other conditions can also lead to elevated PSA levels.
Because of the ease of urine collection and the fact that prostate cells are directly released into the urethra through prostatic ducts after DRE or prostate massage, urine is attractive for noninvasive biomarker testing. Several biomarker candidates for prostate cancer have been identified in urine, including prostate cancer cells, DNA, RNA, proteins, exosomes and other small molecules. Several studies have examined panels of urinary biomarkers for the detection of prostate cancer and currently four urinary tests are commercially available [8].
-
Breast cancer
Despite the widespread use of mammography screening for breast cancer, its effectiveness varies with age. Urine can contain biomarkers such as proteins as well as long-coding RNAs for breast cancer detection [9]. Novosanis' URODETECT project (co-funded by VLAIO, Belgium) also focuses on urine as a potential sample type to improve cancer detection for prostate and breast cancers [10].
-
Bladder cancer
Early detection of bladder cancer can improve chances of survival. However, current detection methods are costly, invasive and can be uncomfortable for the patient. Given the function of the bladder and its proximity to the urinary tract, urine as a sample type is promising as it contains a reliable source of cancer biomarkers. Several biomarker candidates, including DNA, RNA, and proteins, have been identified in urine for bladder cancer detection and monitoring [11].
More work needs to be done to better understand if variables such as urine collection, urine fractions, as well as whether storage, and shipping conditions influence sample quality and impact biomarker detection [11].
Is yellow the new red?
Urine as a liquid biopsy offers huge potential for cancer biomarker testing. Given the challenges with tissue and blood, we believe urine offers a big opportunity in cancer biomarker testing, enabling cancer detection and follow-up in a non-invasive and easy way [6]. We are excited to be part of this space and offer an alternative, yet effective sample type that could help prevent the next global health crisis.
- Fagundes TP, Albuquerque RM, Miranda DLP, Landeiro LCG, Ayres GSF, Correia CCE, NogueiraRodrigues A. Dealing with cancer screening in the COVID-19 era. Rev Assoc Med Bras (1992). 2021 Aug 13;67Suppl 1(Suppl 1):86-90. doi: 10.1590/1806-9282.67.Suppl1.20200889. PMID: 34406299.
- https://www.weforum.org/agenda/2022/01/cancer-health-crisis-oncology-astrazeneca/
- Kang YJ, Baek JM, Kim YS, Jeon YW, Yoo TK, Rhu J, Shin CH, Cho S, Choi H, Oh SJ. Impact of the COVID-19 Pandemic on the Diagnosis and Surgery of Breast Cancer: A Multi-Institutional Study. J Breast Cancer. 2021 Dec;24(6):491-503. https://doi.org/10.4048/jbc.2021.24.e55
- https://news.cancerresearchuk.org/2020/06/01/impact-of-coronavirus-on-cancer-services-revealedover-2-million-people-waiting-for-screening-tests-and-treatments/
- https://www.standaard.be/cnt/dmf20220112_98005734
- Urine as a liquid biopsy - is it the holy grail? https://novosanis.com/sites/default/files/poster/pdf/White%20Paper%20Urine%20as%20a%20liquid%20biopsy%202019-9A_0.pdf?1645106801
- https://novosanis.com/blog/urine-preferred-sample-type-hpv-detection-cervical-cancer-screening
- Multi-omics and urine as same type – a promising approach to detect and monitor prostate cancer https://novosanis.com/sites/default/files/poster/pdf/White%20Paper_Urine%20as%20a%20sample%20 type%20for%20multi-omic%20cancer%20research_Version2021-06A-EN.pdf?1645106801
- https://novosanis.com/infographics/breast-cancer-awareness-month-2020
- https://novosanis.com/urodetect
- Urine as an emerging liquid biopsy for bladder cancer biomarkers https://novosanis.com/sites/default/files/poster/pdf/Urine%20as%20an%20emerging%20liquid%20biopsy%20for%20bladder%20cancer%20biomarkers.pdf?1645106801
