Urine - A preferred sample type for HPV detection for cervical cancer screening
You are here

Background:
Cervical cancer can be prevented through vaccination and screening; however, it remains the fourth most common cancer type in women, with over 500,000 cases detected in 2018 (1). Most cases of cervical cancer have been linked to types of high-risk Human Papillomavirus (hrHPV), a common sexually transmitted infection (1).
Regular cytology-based screening for cervical cancer via a Pap smear, which look for abnormal cervical cells, has directly shown to improve incidence and mortality rates (2,3). However, screening up-take greatly varies from region to region.

In developed countries, average screening coverage is approximately 63%. Austria and Luxembourg show highest screening participation with about 80% of eligible women reporting a pelvic exam and a Pap smear within the last 3 years. In developing countries, screening rates can drop dramatically, with average screening of 19%. Bangladesh, Ethiopia, and Myanmar show the lowest screening coverage of about 1% or less (2).
Why is screening coverage low in many areas:
Despite the benefits of screening, many women are often reluctant to undergo a Pap smear due to several barriers (3). Smears are invasive and are associated with physical discomfort. Additionally, religion and culture can also influence decision of testing. Women have also listed lack of time, inconvenient clinic hours or lack of transportation as reasons for avoiding regular testing (3,4).
Potential of self-sampling:
Given the challenges women face with a Pap smear, ways to improve screening are required. Self-collection techniques have been recommended, which offer better acceptance (5). These methods can significantly improve screening coverage and reach those who do not routinely attend screening programs (6). A meta-analysis reported an overall 2.14 fold increase in screening coverage due to the use of self-collected samples (3), highlighting the impact self-sampling methods could have in cervical cancer screening uptake.

Urine as a sample type:
One method of self-sampling is through urine collection. Women have shown a significantly higher preference to urine as a sample type for hrHPV testing in comparison to brush-based cervical-vaginal self-sampling methods (7,8). Around 85% of participants reported no concerns with urine collection. Alternatively, 16% of women had concerns with performing brush-based methods, and were worried about sample contamination or whether enough specimen had been collected (8).
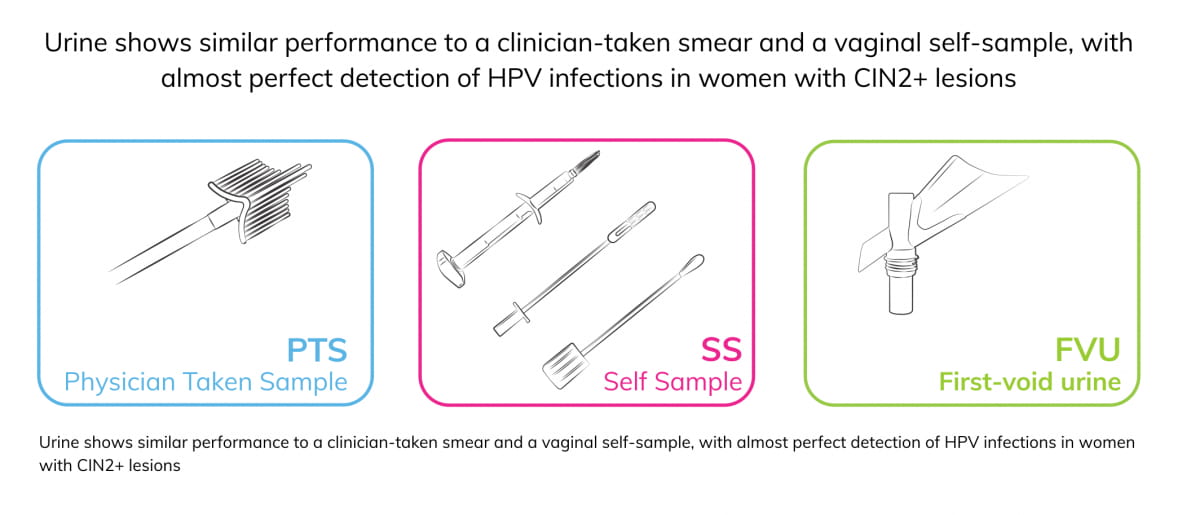
Urine is also preferred as it non-invasive and suitable for home-collection (7,9). Similar sensitivities have been observed between urine and clinician-taken smears as well as brush-based self-sampling for detecting high-grade precancer lesions i.e. CIN2+ (cervical intraepithelial neoplasia grade 2+) (9), highlighting the potential of urine as a sample type.
Colli-Pee® - An innovative urine collection solution:
Interestingly, urine fractions are not the same. First-void urine (initial urine flow) contain higher concentrations of HPV DNA than midstream urine (10,11). As this fraction of urine contains the most cervicovaginal cells, mucus and debris (10), it is critical to capture the appropriate segment for improved sensitivity. Collecting a first-void urine sample with a standard urine cup can be awkward, messy and inconvenient for the user (8). For this reason, Novosanis developed Colli-Pee®, which allows for standardized, volumetric collection of first-void urine. The device architecture also enables immediate mixing of the sample with a urine preservative, improving the stability of the sample (11). Colli-Pee®, prefilled with non-toxic Novosanis proprietary UCM is recommended for HPV testing. Colli-Pee®, usability data show more accurate and volumetric collection of first-void urine with ColliPee® than a traditional urine cup, and that users find the device easy or very easy to use (12,13).
Conclusion:
Current cervical cancer screening methods have drawbacks, resulting in poor attendance. Women would prefer easy, and non-invasive techniques that are not clinician dependent. Urine as a sample type is well-accepted, and given its ability to detect HPV, has the potential to reach a wider population, especially women who do not participate in screening.
References
(1) Cervical cancer – World Health Organization - https://www.who.int/health-topics/cervical-cancer#tab=tab_1
(2) Gakidou E, Nordhagen S, Obermeyer Z. Coverage of cervical cancer screening in 57 countries: low average levels and large inequalities. PLoS Med. 2008 Jun 17;5(6):e132. doi: 10.1371/journal.pmed.0050132. PubMed PMID: 18563963; PubMed Central PMCID: PMC2429949.
(3) Racey CS, Withrow DR, Gesink D. Self-collected HPV testing improves participation in cervical cancer screening: a systematic review and meta-analysis. Can J Public Health. 2013 Feb 11;104(2):e159-66. Review. PubMed PMID: 23618210; PubMed Central PMCID: PMC6973997.
(4) Sarah Wilding , Sarah Wighton , Daisy Halligan , Robert West , Mark Conner & Daryl B. O’Connor (2020) What factors are most influential in increasing cervical cancer screening attendance? An online study of UK-based women1 , Health Psychology and Behavioral Medicine,8:1, 314-328, DOI: 10.1080/21642850.2020.1798239
(5) Ducancelle A, Reiser J, Pivert A, Le Guillou-Guillemette H, Le Duc-Banaszuk AS, Lunel-Fabiani F. Home-based urinary HPV DNA testing in women who do not attend cervical cancer screening clinics. J Infect. 2015 Sep;71(3):377-84. doi: 10.1016/j.jinf.2015.05.001. Epub 2015 May 9. PubMed PMID: 25964233.
(6) Arbyn M, Peeters E, Benoy I, Vanden Broeck D, Bogers J, De Sutter P, Donders G, Tjalma W, Weyers S, Cuschieri K, Poljak M, Bonde J, Cocuzza C, Zhao FH, Van Keer S, Vorsters A. VALHUDES: A protocol for validation of human papillomavirus assays and collection devices for HPV testing on self-samples and urine samples. J Clin Virol. 2018 Oct;107:52-56. doi: 10.1016/j.jcv.2018.08.006. Epub 2018 Aug 22. PubMed PMID: 30195193.
(7) Van Keer S, Pattyn J, Tjalma WAA, Van Ostade X, Ieven M, Van Damme P, Vorsters A. First-void urine: A potential biomarker source for triage of high-risk human papillomavirus infected women. Eur J Obstet Gynecol Reprod Biol. 2017 Sep;216:1-11. doi: 10.1016/j.ejogrb.2017.06.036. Epub 2017 Jun 27. Review. PubMed PMID: 28689156.
(8) Rohner E, McGuire FH, Liu Y, Li Q, Miele K, Desai SA, Schmitt JW, Knittel A, Nelson JAE, Edelman C, Sivaraman V, Baker A, Romocki LS, Rahangdale L, Smith JS. Racial and Ethnic Differences in Acceptability of Urine and Cervico-Vaginal Sample Self-Collection for HPVBased Cervical Cancer Screening. J Womens Health (Larchmt). 2020 Jul;29(7):971-979. doi: 10.1089/jwh.2019.8132. Epub 2020 Mar 26. PubMed PMID: 32212991; PubMed Central PMCID: PMC7371548.
(9) Leeman A, Del Pino M, Molijn A, Rodriguez A, Torné A, de Koning M, Ordi J, van Kemenade F, Jenkins D, Quint W. HPV testing in firstvoid urine provides sensitivity for CIN2+ detection comparable with a smear taken by a clinician or a brush-based self-sample: crosssectional data from a triage population. BJOG. 2017 Aug;124(9):1356-1363. doi: 10.1111/1471-0528.14682. PubMed PMID: 28391609.
(10) Vorsters A, Van den Bergh J, Micalessi I, Biesmans S, Bogers J, Hens A, De Coster I, Ieven M, Van Damme P. Optimization of HPV DNA detection in urine by improving collection, storage, and extraction. Eur J Clin Microbiol Infect Dis. 2014 Nov;33(11):2005-14. doi: 10.1007/s10096-014-2147-2. Epub 2014 Jun 12. PubMed PMID: 24916950.
(11) Pattyn J, Van Keer S, Biesmans S, Ieven M, Vanderborght C, Beyers K, Vankerckhoven V, Bruyndonckx R, Van Damme P, Vorsters A. Human papillomavirus detection in urine: Effect of a first-void urine collection device and timing of collection. J Virol Methods. 2019 Feb;264:23-30. doi: 10.1016/j.jviromet.2018.11.008. Epub 2018 Nov 16. PubMed PMID: 30452931.
(12) Meers N, Donné J, Beyers K, Van den Bossche R, Van Mulder T, Ceccarelli B, Urlings J, Wauben P,De Baetselier I, Crucitti T, Wong J, Iwasiow R, Doukhanine E, MacDonald K, Vankerckhoven V. Standardized and volumetric collection of first-void urine for detection of STIs and HPV: A comparison between Colli-Pee® and a standard urine cup. December 2019
(13) De Baetselier I, Smet H, Abdellati S, De Deken B, Cuylaerts V, Reyniers T, Vuylsteke B, Crucitti T. Evaluation of the 'Colli-Pee', a firstvoid urine collection device for selfsampling at home for the detection of sexually transmitted infections, versus a routine clinicbased urine collection in a one-to-one comparison study design: efficacy and acceptability among MSM in Belgium. BMJ Open. 2019 Apr 3;9(4):e028145. doi: 10.1136/bmjopen-2018-028145. PubMed PMID: 30948618; PubMed Central PMCID: PMC6500257.
