Combat cervical cancer through screening
You are here

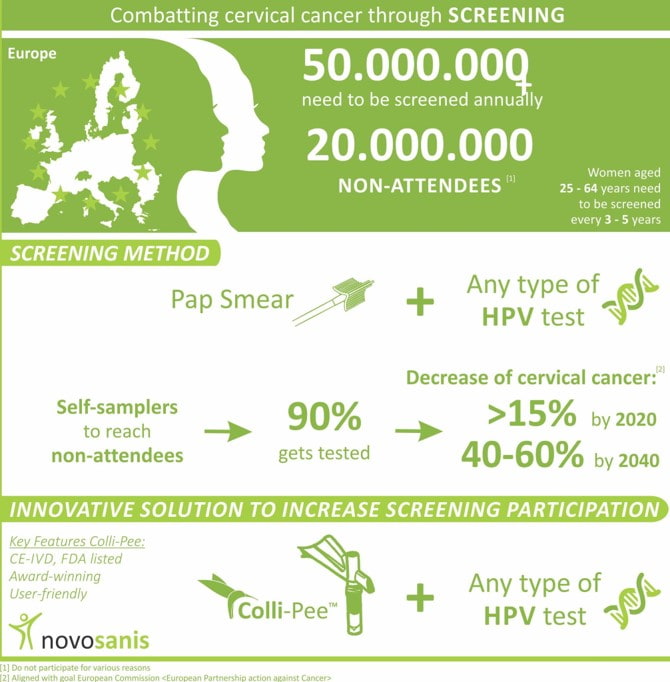
Human Papillomavirus (HPV) infection is the most common sexually transmitted infection (STI), affecting both men and women. The virus can lead to fertility problems as well as many forms of cancer, specifically, cervical cancer (3). Strains of HPV, in particular HPV16 and 18 have been linked to most cervical cancer cases. This cancer type usually does not present any symptoms until it reaches an advanced stage, making early detection and treatment challenging. While women are invited to have a cervical sample through public screening programs, participation remains low in many areas (1).
Over the last few years, Novosanis has invested time and research to see the possibilities of HPV detection in first-void urine collected through our patented and award-winning medical device, Colli-Pee. The tool allows effective capture of the first 20ml of urine, which contains higher concentrations of the virus. Other benefits of first-void urine sampling are it is quick, non-invasive and can easily be performed independently by women.
Alongside HPV, another common STI is Chlamydia. The disease, which is caused by a bacterium called Chlamydia Trachomatis (CT) is highly contagious and can be transmitted between individuals through vaginal, anal or oral sex. Symptoms of Chlamydia are not easily evident; however, if unmanaged, it can cause other health issues. In women, it can lead to an infection of the urethra, inflammation of the cervix, pelvic inflammatory disease and in some cases, infertility.
Research shows how Chlamydia bacteria can also increase the risk of cervical cancer. The infection-associated inflammatory response may damage cell DNA, resulting in genetic instability and eventual tumour development. The infection could also increase the potential of cervical cancer by allowing easier entry of HPV as well as creating a favourable environment for HPV persistence (4).
Studies have demonstrated the ability to detect CT in female urine (2). While more research needs to be performed to evaluate its potential and benefits in comparison to other methods, the possibility to detect both CT and HPV in the same urine sample is promising and offers great convenience for testing.
References:
(1). Ducancelle A, Reiser J, Pivert A, Le Guillou-Guillemette H, Le Duc-Banaszuk AS, Lunel-Fabiani F. Home-based urinary HPV DNA testing in women who do not attend cervical cancer screening clinics. J Infect. 2015 Sep;71(3):377-84. doi: 10.1016/j.jinf.2015.05.001. Epub 2015 May 9. PubMed PMID: 25964233.
(2) Frati ER, Fasoli E, Martinelli M, Colzani D, Bianchi S, Carnelli L, Amendola A, Olivani P, Tanzi E. Sexually Transmitted Infections: A Novel Screening Strategy for Improving Women's Health in Vulnerable Populations. Int J Mol Sci. 2017 Jun 20;18(6). doi: 10.3390/ijms18061311. PubMed PMID: 28632191; PubMed Central PMCID: PMC5486132.
(3) Souho T, Benlemlih M, Bennani B. Human papillomavirus infection and fertility alteration: a systematic review. PLoS One. 2015 May 18;10(5):e0126936. doi: 10.1371/journal.pone.0126936. eCollection 2015. Review. PubMed PMID: 25992782; PubMed Central PMCID: PMC4436317.
(4) Zhu H, Shen Z, Luo H, Zhang W, Zhu X. Chlamydia Trachomatis Infection-Associated Risk of Cervical Cancer: A Meta-Analysis. Medicine (Baltimore). 2016 Mar;95(13):e3077. doi: 10.1097/MD.0000000000003077. Review. PubMed PMID: 27043670; PubMed Central PMCID: PMC4998531.
